Barium hypochlorite
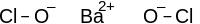 | |
| Identifiers | |
|---|---|
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | Ba(OCl)2 |
| Molar mass | 240.232 g/mol |
| Appearance | white to colorless crystals |
| Melting point | 235 °C (455 °F; 508 K) (decomposes) |
Solubility in water | reacts |
| Related compounds | |
Other anions |
|
Other cations |
|
Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Barium hypochlorite is a chemical compound with the chemical formula Ba(OCl)2 (also written as Ba(ClO)2). It is the barium salt of hypochlorous acid. It consists of barium cations Ba2+ and hypochlorite anions −OCl.
Applications
It is used as a bleaching agent of textiles, paper and pulp; in the decontamination of explosives; as an antiseptic; as an ingredient to make chloropicrin.
References
- Chemical Encyclopedia / Editorial Board.: Knunyants IL and others. - M .: Soviet Encyclopedia, 1988. - T. 1. - 623 p.
- Handbook of chemical / Editorial Board.: Nikolsky BP and others. - 3rd ed., Rev. - L. : Chemistry, 1971. - T. 2. - 1168 s
- v
- t
- e
- BaB6
- Ba(BO2)2
- BaBr2
- Ba(BrO3)2
- Ba(CH3CO2)2
- Ba(C5H7O2)2
- Ba(ClO)2
- BaC2
- BaCO3
- BaC2O4
- Ba(ClO3)2
- BaClF
- Ba(ClO4)2
- Ba(CN)2
- BaCl2
- BaCrO4
- BaF2
- BaFeO4
- BaFe2O4
- BaH2
- BaI2
- Ba(IO3)2
- BaMnO4
- Ba(MnO4)2
- Ba(N3)2
- Ba(NO2)2
- Ba(NO3)2
- BaO
- BaO2
- Ba(OH)2
- Ba(PO3)2
- BaS
- BaSe
- BaSeO4
- Ba(SCN)2
- BaSO3
- BaSO4
- BaRuO3
- BaSnO3
- BaTiO3
- Ba2TiO4
- BaWO4
- BaZnGa
- Sr2Ba1-xNb2O6
- YBa2Cu3O7-x










