Sec-Butyllithium
 | |
 | |
| Names | |
|---|---|
| IUPAC name sec-Butyllithium | |
| Systematic IUPAC name Butan-2-yllithium | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
Beilstein Reference | 3587206 |
| ChemSpider |
|
| ECHA InfoCard | 100.009.026 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C4H9Li |
| Molar mass | 64.06 g·mol−1 |
| Acidity (pKa) | 51 |
| Hazards | |
| Safety data sheet (SDS) | Fisher MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |
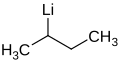
sec-Butyllithium is an organometallic compound with the formula CH3CHLiCH2CH3, abbreviated sec-BuLi or s-BuLi. This chiral organolithium reagent is used as a source of sec-butyl carbanion in organic synthesis.[1]
Synthesis
sec-BuLi can be prepared by the reaction of sec-butyl halides with lithium metal:[2]

Properties
Physical properties
sec-Butyllithium is a colorless viscous liquid.[1][3] Using mass spectrometry, it was determined that the pure compound has a tetrameric structure.[4] It also exists as tetramers when dissolved in organic solvents such as benzene, cyclohexane or cyclopentane.[3] The cyclopentane solution has been detected with 6Li-NMR spectroscopy to have a hexameric structure at temperatures below −41 °C.[5] In electron-donating solvents such as tetrahydrofuran, there exists an equilibrium between monomeric and dimeric forms.[6]
Chemical properties
The carbon-lithium bond is highly polar, rendering the carbon basic, as in other organolithium reagents. Sec-butyllithium is more basic than the primary organolithium reagent, n-butyllithium. It is also more sterically hindered. sec-BuLi is employed for deprotonations of particularly weak carbon acids where the more conventional reagent n-BuLi is unsatisfactory. It is, however, so basic that its use requires greater care than for n-BuLi. For example diethyl ether is attacked by sec-BuLi at room temperature in minutes, whereas ether solutions of n-BuLi are stable.[1]
The compound decomposes slowly at room temperature and more rapidly at higher temperatures, giving lithium hydride and a mixture of butenes.[7][8]
Applications
Many transformations involving sec-butyllithium are similar to those involving other organolithium reagents.
In combination with sparteine as a chiral auxiliary, sec-butyllithium is useful in enantioselective deprototonations.[9] It is also effective for lithiation of arenes.[10]
References
- ^ a b c Ovaska, T. V. (2001). "s-Butyllithium". Encyclopedia of Reagents for Organic Synthesis. New York: John Wiley & Sons. doi:10.1002/047084289X.rb397. ISBN 0471936235..
- ^ Hay, D. R.; Song, Z.; Smith, S. G.; Beak, P. (1988). "Complex-induced proximity effects and dipole-stabilized carbanions: kinetic evidence for the role of complexes in the α-lithiations of carboxamides". J. Am. Chem. Soc. 110 (24): 8145–8153. doi:10.1021/ja00232a029.
- ^ a b Wietelmann, Ulrich; Bauer, Richard J. (2000-06-15), "Lithium and Lithium Compounds", in Wiley-VCH Verlag GmbH & Co. KGaA (ed.), Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. a15_393, doi:10.1002/14356007.a15_393, ISBN 978-3-527-30673-2, retrieved 2022-05-07
- ^ Plavsic, D.; Srzic, D.; Klasinc, Leo (1986). "Mass spectrometric investigations of alkyllithium compounds in the gas phase". The Journal of Physical Chemistry. 90 (10): 2075–2080. doi:10.1021/j100401a020. ISSN 0022-3654.
- ^ Fraenkel, Gideon; Henrichs, Mark; Hewitt, Michael; Su, Biing Ming (1984). "Structure and dynamic behavior of a chiral alkyllithium compound: carbon-13 and lithium-6 NMR of sec-butyllithium". Journal of the American Chemical Society. 106 (1): 255–256. doi:10.1021/ja00313a052. ISSN 0002-7863.
- ^ Bauer, Walter.; Winchester, William R.; Schleyer, Paul von R. (1987-11-01). "Monomeric organolithium compounds in tetrahydrofuran: tert-butyllithium, sec-butyllithium, supermesityllithium, mesityllithium, and phenyllithium. Carbon-lithium coupling constants and the nature of carbon-lithium bonding". Organometallics. 6 (11): 2371–2379. doi:10.1021/om00154a017. ISSN 0276-7333.
- ^ Glaze, William H.; Lin, Jacob; Felton, E. G. (1965). "The Thermal Decomposition of sec-Butyllithium". The Journal of Organic Chemistry. 30 (4): 1258–1259. doi:10.1021/jo01015a514. ISSN 0022-3263.
- ^ Glaze, William H.; Lin, Jacob; Felton, E. G. (1966). "The Pyrolysis of Unsolvated Alkyllithium Compounds". The Journal of Organic Chemistry. 31 (8): 2643–2645. doi:10.1021/jo01346a044. ISSN 0022-3263.
- ^ Crépy, Karen V. L.; Imamoto, Tsuneo (2005). "Preparation of (S,S)-1,2-bis(tert-Butylmethylphosphino)ethane ((S,S)-t-bu-bisp*) as a Rhodium Complex". Organic Syntheses. 82: 22. doi:10.15227/orgsyn.082.0022.
- ^ Wang, X.; de Silva, S. O.; Reed, J. N.; Billadeau, R.; Griffen, E. J.; Chan, A.; Snieckus, V. (1995). "7-Methoxyphthalide". Org. Synth. 72: 163. doi:10.15227/orgsyn.072.0163.
- v
- t
- e
- Li2
- LiAlCl4
- Li1+xAlxGe2−x(PO4)3
- LiAlH4
- LiAlO2
- LiAl1+xTi2−x(PO4)3
- LiAs
- LiAsF6
- Li3AsO4
- LiAt
- Li[AuCl4]
- LiB(C2O4)2
- LiB(C6F5)4
- LiBF4
- LiBH4
- LiBO2
- LiB3O5
- Li2B4O7
- Li2TiF6
- Li2ZrF6
- Li2B4O7·5H2O
- LiBSi2
- LiBr
- LiBr·2H2O
- LiBrO
- LiBrO2
- LiBrO3
- LiBrO4
- Li2C2
- LiCF3SO3
- CH3CH(OH)COOLi
- LiC2H2ClO2
- LiC2H3IO2
- Li(CH3)2N
- LiCHO2
- LiCH3O
- LiC2H5O
- LiCN
- Li2CN2
- LiCNO
- Li2CO3
- Li2C2O4
- LiCl
- LiCl·H2O
- LiClO
- LiFO
- LiClO2
- LiClO3
- LiClO4
- LiCoO2
- Li2CrO4
- Li2CrO4·2H2O
- Li2Cr2O7
- CsLiB6O10
- LiD
- LiF
- Li2F
- LiF4Al
- Li3F6Al
- FLiBe
- LiFePO4
- FLiNaK
- LiGaH4
- Li2GeF6
- Li2GeO3
- LiGe2(PO4)3
- LiH
- LiH2AsO4
- Li2HAsO4
- LiHCO3
- Li3H(CO3)2
- LiH2PO3
- LiH2PO4
- LiHSO3
- LiHSO4
- LiHe
- LiI
- LiIO
- LiIO2
- LiIO3
- LiIO4
- Li2IrO3
- Li7La3Zr2O12
- LiMn2O4
- Li2MoO4
- Li0.9Mo6O17
- LiN3
- Li3N
- LiNH2
- Li2NH
- LiNO2
- LiNO3
- LiNO3·H2O
- Li2N2O2
- LiNa
- Li2NaPO3
- LiNaNO2
- LiNbO3
- Li2NbO3
- LiO−
- LiO2
- LiO3
- Li2O
- Li2O2
- LiOH
- Li3P
- LiPF6
- Li3PO4
- Li2HPO3
- Li2HPO4
- Li3PO3
- Li3PO4
- Li2Po
- Li2PtO3
- Li2RuO3
- Li2S
- LiSCN
- LiSH
- LiSO3F
- Li2SO3
- Li2SO4
- Li[SbF6]
- Li2Se
- Li2SeO3
- Li2SeO4
- LiSi
- Li2SiF6
- Li4SiO4
- Li2SiO3
- Li2Si2O5
- LiTaO3
- Li2Te
- LiTe3
- Li2TeO3
- Li2TeO4
- Li2TiO3
- Li4Ti5O12
- LiTi2(PO4)3
- LiVO3·2H2O
- Li3V2(PO4)3
- Li2WO4
- LiYF4
- LiZr2(PO4)3
- Li2ZrO3
- Hemolithin (extraterrestrial protein)
- Organolithium reagents
- CH3COOLi
- C4H6LiNO4
- LiC2F6NO4S2
- LiN(SiMe3)2
- Li3C6H5O7
- C5H5Li
- LiN(C3H7)2
- (C6H5)2PLi
- C18H35LiO3
- C6H13Li
- C4H9Li
- CH3CHLiCH2CH3
- (CH3)3CLi
- C12H28BLi
- CH3Li
- Li+C10H8−
- C5H11Li
- C5H3LiN2O4
- C6H5Li
- LiC2CH3
- LiO2C(CH2)16CH3
- C4H5LiO4
- LiEt3BH
- LiOC(CH3)3
- C9H18LiN
- LiC2H3 Vinyllithium
- LiC
11H
23COO
- Amblygonite
- Berezanskite
- Brannockite
- Cryolithionite
- Darapiosite
- Darrellhenryite
- Elbaite
- Fluorine Elbaite
- Fluor-liddicoatite
- Emeleusite
- Eucryptite LiAlSiO4
- Faizievite
- Hectorite
- Hsianghualite
- Jadarite LiNaSiB3O7OH
- Keatite Li(AlSi2O6)
- Kunzite
- Lavinskyite
- Lepidolite
- Lithiophilite LiMnPO4
- Lithiophosphate Li3PO4
- Manandonite
- Manganoneptunite
- Nambulite
- Neptunite
- Olympite
- Petalite LiAlSi4010
- Pezzottaite Cs(Be2Li)Al2Si6O18
- Rossmanite
- Saliotite
- Sogdianite
- Spodumene LiAl(SiO3)2
- Sugilite
- Tiptopite
- Tourmaline
- Triphylite LiFePO4
- Zabuyelite Li2CO3
- Zektzerite
- Zinnwaldite
- LixBey
- HLiHe+
- LiFHeO
- LiHe2
- (HeO)(LiF)2
- La2/3-xLi3xTiO3He
- Aluminium–lithium alloys
- Heteroatom-promoted lateral lithiation
- LB buffer
- Lithium atom
- Lithium medication
- LiNixCoyAlzO2
- LiNixMnyCozO2
- Lithium soap
- Lithium Triangle
- Lucifer yellow
- Magnesium–lithium alloys
- NASICON
- Environmentally friendly red light flare












