Devazepid
 | |||
| (IUPAC) ime | |||
|---|---|---|---|
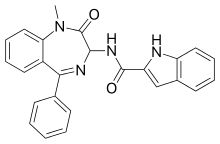
| N-(1-methyl- 2-oxo- 5-phenyl- 3H-1,4-benzodiazepin-3-yl)- 1H-indole -2-carboxamide | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 103420-77-5 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 59751 | ||
| KEGG[3] | D02693  Y Y | ||
| ChEMBL[4] | CHEMBL153326  Y Y | ||
| Hemijski podaci | |||
| Formula | C25H20N4O2 | ||
| Mol. masa | 408.452 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | |||
Devazepid (L-364,718, MK-329) je lek koji je strukturni derivat benzodiazepinske familije, ali se po dejstvu veoma razlikuje od većine benzodiazepina. On nema afiniteta za GABAA receptore i umesto toga deluje kao holecistokininski antagonist koji je selektivan za CCKA tip receptora.[5] On povećava apetit i ubrzava želudačano pražnjenje,[6][7] i smatra se da on potencijalno može da služi kao lek za niz gastrointestinalnih problema, među kojima su dispepsija, gastropareza i gastroezofagealna refluksna bolest.[8] On je u širokoj upotrebi u naučnim istraživanjima CCKA receptora.[9][10][11]

Reference
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519. edit
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594. edit
- ↑ Hill DR, Woodruff GN. Differentiation of central cholecystokinin receptor binding sites using the non-peptide antagonists MK-329 and L-365,260. Brain Research. 1990 Sep 3;526(2):276-83. PMID 2257485
- ↑ Cooper SJ, Dourish CT. Multiple cholecystokinin (CCK) receptors and CCK-monoamine interactions are instrumental in the control of feeding. Physiology and Behaviour. 1990 Dec;48(6):849-57. PMID 1982361
- ↑ Cooper SJ, Dourish CT, Clifton PG. CCK antagonists and CCK-monoamine interactions in the control of satiety. American Journal of Clinical Nutrition. 1992 Jan;55(1 Suppl):291S-295S. PMID 1728842
- ↑ Scarpignato C, Varga G, Corradi C. Effect of CCK and its antagonists on gastric emptying. Journal of Physiology Paris. 1993;87(5):291-300. PMID 8298606
- ↑ Weller A. The ontogeny of postingestive inhibitory stimuli: examining the role of CCK. Developmental Psychobiology. 2006 Jul;48(5):368-79. PMID 16770766
- ↑ Savastano DM, Covasa M. Intestinal nutrients elicit satiation through concomitant activation of CCK(1) and 5-HT(3) receptors. Physiology and Behaviour. 2007 Oct 22;92(3):434-42. PMID 17531277
- ↑ Evans, B. E.; Rittle, K. E.; DiPardo, R. M.; Freidinger, R. M.; Whitter,W. L.; People,W. T.; Lendell, G. F.; Veber, D. F.; Anderson, P. S.; Chang, R. S. L.; Lotti, V. J.; Cerino, D. J.; Chen, T. B.; Kling, P. J.; Kunkel, K. A.; Springer, J. P.; Hirschfield, J. (1988). „Methods for drug discovery: development of potent, selective, orally effective cholecystokinin antagonists”. J. Med. Chem. 31: 2235.
Spoljašnje veze
 | Portal Medicina |
 | Portal Hemija |
- p
- r
- u
Neuropeptidni ligandi
CCKA | Agonisti: Holecistokinin • CCK-4 Antagonisti: Asperlicin • Proglumid • Lorglumid • Devazepid • Deksloksiglumid |
|---|---|
CCKB |
CRF1 | |
|---|---|
CRF2 | Agonisti: Kortikotropin-oslobađajući hormon |
GAL1 | |
|---|---|
GAL2 | |
GAL3 |
MCH1 | Agonisti: Melanin-koncentrirajući hormon Antagonisti: ATC-0175 • GW-803,430 • NGD-4715 • SNAP-7941 • SNAP-94847 |
|---|---|
MCH2 | Agonisti: Melanin-koncentrirajući hormon |
MC1 | |
|---|---|
MC2 | |
MC3 | |
MC4 | |
MC5 | Agonisti: alfa-MSH • Melanotan II |
Y1 | |
|---|---|
Y2 | |
Y4 | |
Y5 |
NTS1 | |
|---|---|
NTS2 |
vidi Opioidi
OX1 | |
|---|---|
OX2 |
Agonisti: Karbetocin • Demoksitocin • Oksitocin • WAY-267,464
Antagonisti: Atosiban • L-371,257 • L-368,899
Antagonisti: Atosiban • L-371,257 • L-368,899
NK1 | Agonisti: Supstanca P Antagonisti: Aprepitant • Befetupitant • Kasopitant • CI-1021 • CP-96,345 • CP-99,994 • CP-122,721 • Dapitant • Ezlopitant • FK-888 • Fosaprepitant • GR-203,040 • GW-597,599 • HSP-117 • L-733,060 • L-741,671 • L-743,310 • L-758,298 • Lanepitant • LY-306,740 • Maropitant • Netupitant • NKP-608 • Nolpitantium • Orvepitant • RP-67,580 • SDZ NKT 343 • Vestipitant • Vofopitant |
|---|---|
NK2 | |
NK3 |
V1A | Agonisti: Dezmopresin • Felipresin • Ornipresin • Terlipresin • Vazopresin Antagonisti: Konivaptan • Demeklociklin • Relkovaptan |
|---|---|
V1B | |
V2 | Agonisti: Dezmopresin • Ornipresin • Vazopresin Antagonisti: Konivaptan • Demeklociklin • Liksivaptan • Mozavaptan • Satavaptan • Tolvaptan |







