Clentiazem
 | |
| Names | |
|---|---|
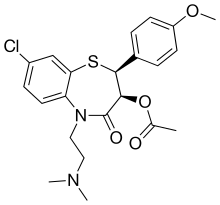
| Preferred IUPAC name (2S,3S)-8-Chloro-5-[2-(dimethylamino)ethyl]-2-(4-methoxyphenyl)-4-oxo-2,3,4,5-tetrahydro-1,5-benzothiazepin-3-yl acetate | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C22H25ClN2O4S |
| Molar mass | 448.9629 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Clentiazem is a calcium channel blocker.[1]
It is a chlorine derivative of diltiazem.[2]
References
- ^ Giasson S, Garceau D, Homsy W, Dumont L (October 1995). "Pharmacodynamics and pharmacokinetics of clentiazem and diltiazem in closed-chest anesthetized dogs". Cardiovasc Drugs Ther. 9 (5): 685–92. doi:10.1007/BF00878551. PMID 8573551. S2CID 37503776.
- ^ Dagenais F, Hollmann C, Buluran J, Cartier R (October 1995). "Clentiazem and diltiazem preserve endothelium-dependent relaxation following global rat heart ischemia". Can J Cardiol. 11 (9): 816–22. PMID 7585280.
- v
- t
- e
Ion channel modulators
| VGKCsTooltip Voltage-gated potassium channels |
| ||||
|---|---|---|---|---|---|
| IRKsTooltip Inwardly rectifying potassium channel |
| ||||
| KCaTooltip Calcium-activated potassium channel |
| ||||
| K2PsTooltip Tandem pore domain potassium channel |
|
| VGSCsTooltip Voltage-gated sodium channels |
| ||||
|---|---|---|---|---|---|
| ENaCTooltip Epithelial sodium channel |
| ||||
| ASICsTooltip Acid-sensing ion channel |
|
| CaCCsTooltip Calcium-activated chloride channel |
| ||||
|---|---|---|---|---|---|
| CFTRTooltip Cystic fibrosis transmembrane conductance regulator |
| ||||
| Unsorted |
|
| TRPsTooltip Transient receptor potential channels |
|
|---|---|
| LGICsTooltip Ligand gated ion channels |
|
See also: Receptor/signaling modulators • Transient receptor potential channel modulators











