Nitrosourea
| |||
| Names | |||
|---|---|---|---|
| IUPAC name Nitrosourea | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
PubChem CID |
| ||
| UNII |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | CH3N3O2 | ||
| Molar mass | 89.054 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Chemical compound
Nitrosourea is both the name of a molecule, and a class of compounds that include a nitroso (R-NO) group and a urea.
Examples
Examples include:
- Arabinopyranosyl-N-methyl-N-nitrosourea (Aranose)
- Carmustine (BCNU, BiCNU)
- Chlorozotocin
- Ethylnitrosourea (ENU)
- Fotemustine
- Lomustine (CCNU)
- Nimustine
- N-Nitroso-N-methylurea (NMU)
- Ranimustine (MCNU)
- Semustine
- Streptozocin (Streptozotocin)
Nitrosourea compounds are DNA alkylating agents and are often used in chemotherapy.[1] They are lipophilic and thus can cross the blood–brain barrier, making them useful in the treatment of brain tumors such as glioblastoma multiforme.[2]
-
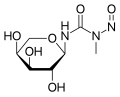 Arabinopyranosyl-N-methyl-N-nitrosourea
Arabinopyranosyl-N-methyl-N-nitrosourea -

-
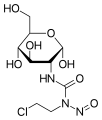
-

-

-

-
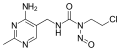
 N-Nitroso-N-methylurea
N-Nitroso-N-methylurea -
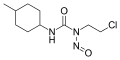
-

-
-

Side effects
Some nitrosoureas (e.g. lomustine) have been associated with the development of interstitial lung disease.[3]
References
- ^ "Antineop". Archived from the original on 2009-03-07. Retrieved 2009-01-24.
- ^ Takimoto CH, Calvo E. "Principles of oncologic pharmacotherapy". in Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ (Eds) Cancer management: a multidisciplinary approach. 11 ed. 2008.
- ^ Tucci E, Verdiani P, Di Carlo S, Sforza V (1986). "Lomustine (CCNU)-induced pulmonary fibrosis". Tumori. 72 (1): 95–8. doi:10.1177/030089168607200114. PMID 3952821. S2CID 33327504.
External links
- Nitrosourea+Compounds at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Diseases Database (DDB): 9052
- v
- t
- e
(M phase)
| Block microtubule assembly | |
|---|---|
| Block microtubule disassembly |
inhibitor
| DNA precursors/ antimetabolites (S phase) |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Topoisomerase inhibitors (S phase) |
| ||||||||
| Crosslinking of DNA (CCNS) |
|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
 | This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e


























